Case Study · B2B SaaS
Somnus
Clinical Study Management Platform
Role
UX/UI Designer
Type
B2B SaaS
Methods
UX & UI Design · User Research · Prototyping
Domain
Clinical Research

Overview
Somnus is built to support clinical study teams in running complex sleep research with confidence and clarity.
The platform brings together site certification, secure file uploads, automated report generation, and real-time study progress monitoring — replacing the patchwork of email threads and spreadsheets that most teams were relying on.
“Multi-site sleep studies suffer from scattered tools, heavy email traffic, and manual spreadsheets. Teams needed a central, user-friendly system to coordinate hundreds of research sites.”
The Challenge
My Role
Led the UX process from discovery through implementation — conducting stakeholder interviews, journey mapping, and workflow analysis with scientists, project managers, and QA leads across multiple research sites.
Phase
01
Discover
Interviews and task analyses with scientists, project managers, and QA leads.
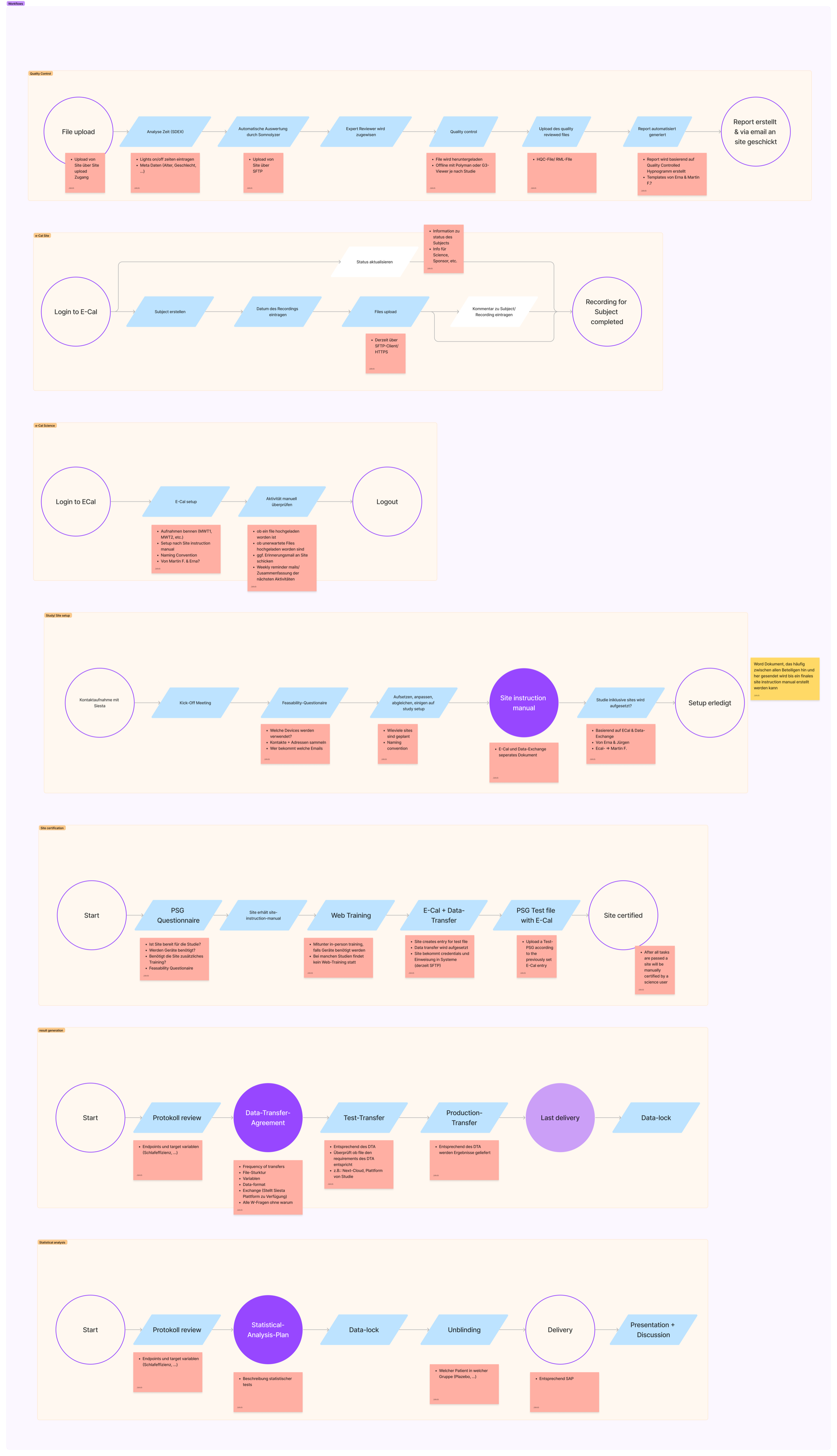
Key findings centred on three recurring pain points: fragmented tools spread across email, spreadsheets, and shared drives; manual communication with no single source of truth; and inconsistent file handling processes that varied by site and researcher.
Phase
02
Define
Framing the core complexity: every site had its own way of doing things.
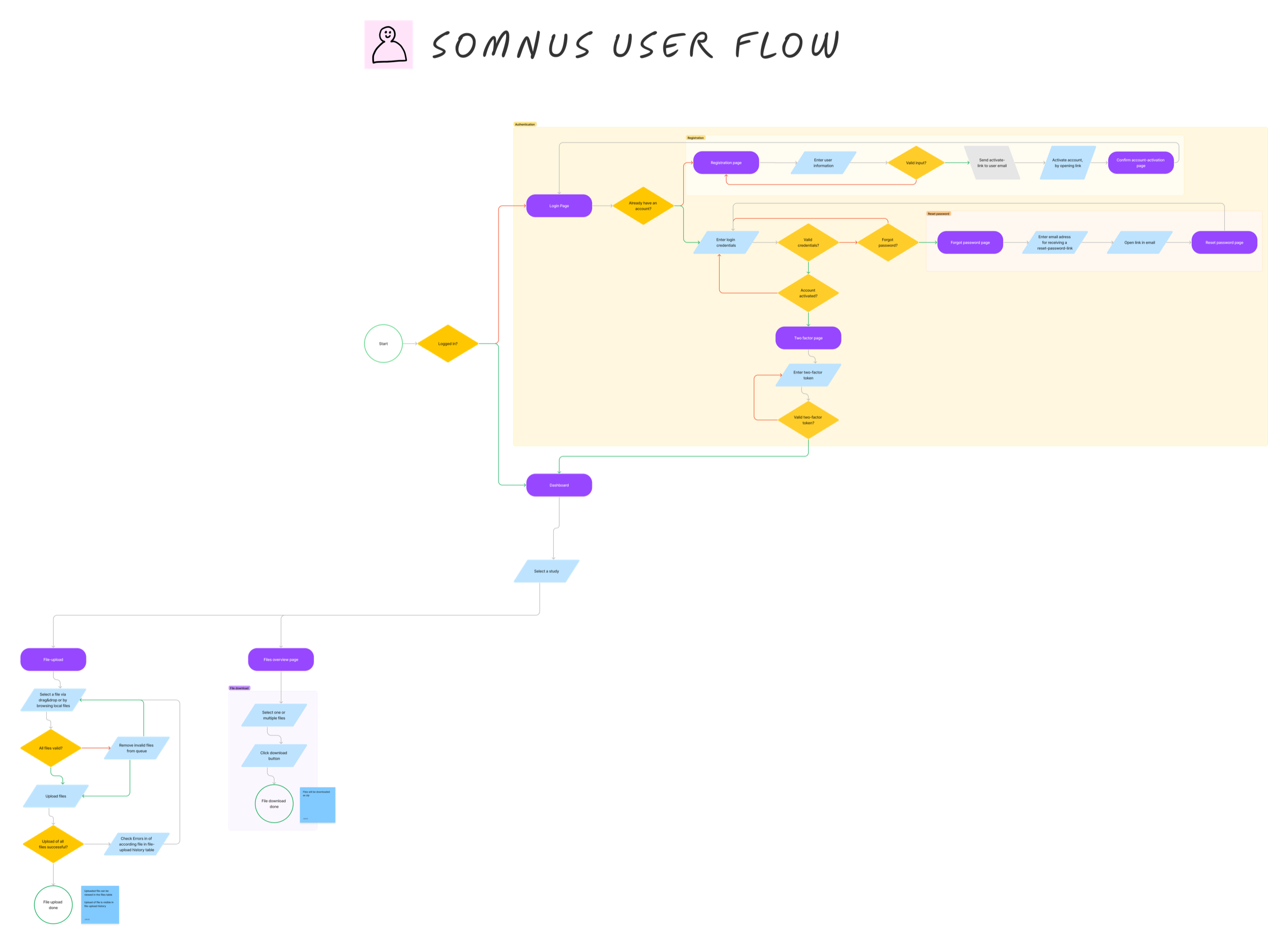
The defining challenge was file upload workflows. Research sites followed different naming conventions, file formats, and preprocessing steps — meaning the same underlying task produced wildly different inputs. Before anything could be designed, this variation needed to be understood and systematised.

Phase
03
Design
User flows and wireframes focused on standardising uploads and introducing validation.
The design phase produced a validation-first upload flow: files are checked against format and naming rules before submission, with clear inline error messaging guiding users to correct issues themselves. This reduced back-and-forth and built trust in the system without requiring manual review at every step.
Phase
04
Deliver
Iterative collaboration with developers throughout sprints.
Rather than a single handoff, delivery happened in close collaboration with the engineering team across iterative sprints. Design decisions were continuously pressure-tested against real-world constraints — database structure, third-party integrations, and regulatory requirements — to ensure the interface reflected what the system could actually do.

Solution
The resulting platform centralises the full study coordination workflow into one system — with a particular focus on reducing the manual overhead that was consuming team capacity.
Site Certification
Structured onboarding flow for research sites with validation checkpoints before study participation.
Secure File Uploads
Standardised upload interface with inline validation — format, naming, and preprocessing rules enforced at submission.
Automated Reporting
Reports generated directly from submitted data, eliminating manual compilation and reducing turnaround time.
Real-time Monitoring
Live study progress dashboard giving coordinators a single view across all active sites.

Outcome
The platform positioned the company as a more modern and efficient player in clinical study operations — moving from a services-based model reliant on manual coordination to a scalable software product teams could operate with confidence.
Next Project
Dog Sitter App
The Ultimate Dog Care Service